Category: Critical Care
Posted: 7/29/2025 by Caleb Chan, MD
Click here to contact Caleb Chan, MD
PEEP is often titrated up along with FiO2 to increase oxygen saturation. Although the potential negative hemodynamic effect of high PEEP is often recognized, it is important to also note that high PEEP can also paradoxically worsen oxygen saturation.
The primary physiologic explanation for this phenomenon in a patient with pulmonary disease is due to the varying impact of PEEP on the intra- vs. extra-alveolar blood vessels. PEEP preferentially distends more normal/compliant lung which causes compression of intra-alveolar vessel at excessively high levels of PEEP. This causes pulmonary blood to be diverted to areas of lower vascular resistance (e.g. consolidated lung which is less distended due to its worsened compliance) and lower VQ matching. Essentially, blood flow to normal/healthy lung is decreased and is instead increased to diseased lung, worsening hypoxemia.
Bottom line:
High PEEP can potentially worsen hypoxemia and should be considered as an etiology for worsening oxygen saturation, particularly when the hypoxemia is out of proportion to the patient’s radiographic findings.
Çoruh B, Luks AM. Positive end-expiratory pressure. When more may not be better. Annals ATS. 2014;11(8):1327-1331.
Category: Critical Care
Posted: 7/23/2025 by William Teeter, MD
Click here to contact William Teeter, MD
Secondary analysis of a multicenter, prospective, observational study ICE-CRASH study in Japan including adult patients admitted with moderate-to-severe accidental hypothermia between 2019 and 2022.
Some structural generalizability (median age 81 years!) issues with this study but well done overall.
Authors undertook some rather complex modeling to predict outcomes related to rapid rewarming, showing that “the rewarming rate and predicted probability of each outcome increased significantly up to 3°C/hr, but when the rewarming rate exceeded 3°C/hr, the predicted probability of each outcome was almost constant.”
Suggests that for those with severe hypothermia that an initially rapid rate of up to 3C/hr is a good target for a ceiling, but above this may be associated with less favorable risk:benefit ratio. Benefit in moderate hypothermia was not as clear.
Conclusion: The mode of rewarming in severe hypothermia should still be based on local protocols and capabilities (e.g. external, intravascular, extracorporeal rewarming) but the rate of rewarming up to 3C/hr is associated with better outcomes.
Category: Critical Care
Keywords: Hypotension, Shock, Mean Arterial Pressure, Vasopressors, Elderly Patients, Geriatrics (PubMed Search)
Posted: 7/15/2025 by Mark Sutherland, MD
Click here to contact Mark Sutherland, MD
Following up Dr. Flint's pearl from the other day, the largest study to date looking at a lower Mean Arterial Pressure (MAP) target in elderly ICU patients is the “65” Trial, published in JAMA in 2020. This trial compared a MAP target of 60-65 to the usual goal of >65, in critically ill patients age 65 and older. It included 2,455 patients in 65 ICUs in the UK, and found no difference between the groups.
Bottom Line: Although most intensivists still target a MAP > 65 regardless of patient age, you do have some evidence to support you if you want to target 60-65 in patients over age sixty-five. However, there are some important limitations (well outlined in the PulmCrit article linked below), and therapy should always be optimized to the patient and markers of end organ perfusion.
Category: Critical Care
Keywords: Sedation, propofol, dexmedetomidine, RASS (PubMed Search)
Posted: 7/8/2025 by Zachary Wynne, MD
Click here to contact Zachary Wynne, MD
The presence of an endotracheal tube by itself does not mandate sedation and many patients require no sedatives while intubated in the ICU. However, patients intubated in the emergency department usually require initial sedation while still paralyzed from RSI. Sedation can also help facilitate procedures and imaging in critically ill patients during initial management.
Current literature has found increased mortality and length of ventilator requirement in oversedated ED patients. The target sedation level for the general population remains a goal RASS (Richmond Agitation-Sedation Scale) of 0 to -1. Society of Critical Care Medicine guidelines from early 2025 recommend dexmedetomidine over propofol as the preferred sedative for light sedation and reducing delirium risk in intubated critically ill patients. A recent trial re-examined other clinical outcomes between these two common sedative agents.
A2B Randomized Clinical Trial - JAMA 2025
Clinical Question: Does alpha 2 adrenergic receptor agonist sedation (dexmedetomidine or clonidine) reduce duration of mechanical ventilation in mechanically ventilated patients compared to a propofol based regimen (usual care)?
Where: 41 UK ICU’s from December 2018 to October 2023
Who: 1438 adults receiving mechanical ventilation for less than 48 hours, receiving propofol and opioid for sedation/analgesia, expected to require mechanical ventilation for greater than 48 hours
Intervention: protocol driven sedation to reach a RASS score of -2 to +1 (either dexmedetomidine, clonidine, or propofol). Of note, propofol could be added to achieve deeper sedation goal if deemed necessary by care team.
Outcomes:
Bottom Line:
While either dexmedetomidine or propofol, with appropriate use of opiates for pain management, are appropriate agents in non-paralyzed mechanically-ventilated patients, propofol may be a more appropriate choice in patients with greater agitation while boarding in the emergency department. However, close attention is needed to avoid the overly deep analgosedation associated with increased mortality. Maintain a goal RASS of 0 to -1 with frequent re-evaluation of your ICU boarders.
Walsh TS, Parker RA, Aitken LM, McKenzie CA, Emerson L, Boyd J, Macdonald A, Beveridge G, Giddings A, Hope D, Irvine S, Tuck S, Lone NI, Kydonaki K, Norrie J, Brealey D, Antcliffe D, Reay M, Williams A, Bewley J, Creagh-Brown B, McAuley DF, Dark P, Wise MP, Gordon AC, Perkins GD, Reade MC, Blackwood B, MacLullich A, Glen R, Page VJ, Weir CJ; A2B Trial Investigators. Dexmedetomidine- or Clonidine-Based Sedation Compared With Propofol in Critically Ill Patients: The A2B Randomized Clinical Trial. JAMA. 2025 Jul 1;334(1):32-45. doi: 10.1001/jama.2025.7200. PMID: 40388916; PMCID: PMC12090071.
Lewis K, Balas MC, Stollings JL, et al. A focused update to the clinical practice guideline for the prevention and management of pain, anxiety, agitation/sedation, delirium, immobility, and sleep disruption in adult patients in the ICU. Crit Care Med. 2025 Mar 1;53(3):e711-e727.
Stephens RJ, Ablordeppey E, Drewry AM, Palmer C, Wessman BT, Mohr NM, Roberts BW, Liang SY, Kollef MH, Fuller BM. Analgosedation Practices and the Impact of Sedation Depth on Clinical Outcomes Among Patients Requiring Mechanical Ventilation in the ED: A Cohort Study. Chest. 2017 Nov;152(5):963-971. doi: 10.1016/j.chest.2017.05.041. Epub 2017 Jun 21. PMID: 28645462; PMCID: PMC5812748.
Category: Critical Care
Posted: 7/1/2025 by Mike Winters, MBA, MD
(Updated: 5/13/2026)
Click here to contact Mike Winters, MBA, MD
When To Initiate RRT in the Critically Ill Patient
Barbar SD, Wald R, Quenot JP. Acute kidney injury: when and how to start renal replacement therapy. Intensive Care Med. 2025;51:1172-1175.
Category: Critical Care
Keywords: ventilation ineffective-trigger double-trigger (PubMed Search)
Posted: 6/24/2025 by Cody Couperus-Mashewske, MD
Click here to contact Cody Couperus-Mashewske, MD
Patient-ventilator dyssynchrony is a sign of a disagreement between the patient's breathing and the ventilator's settings. Recognizing and fixing it is a critical skill to prevent lung injury and improve comfort. Ineffective triggering and double-trigger are two common types of dyssynchrony.
The patient tries to take a breath, but they are too weak to trigger the ventilator. This is the most common type of dyssynchrony. It causes increased work of breathing and discomfort.
Look for a small dip in the pressure waveform and a simultaneous scoop out of the expiratory flow waveform that is not followed by a delivered breath.
Troubleshooting options:
The patient's own breath outlasts the ventilator's set inspiratory time (Ti), causing one patient effort to trigger two stacked breaths. This results in delivery of large tidal volumes, risking lung injury (volutrauma).
Look for two consecutive breaths on the ventilator screen without a full exhalation in between.
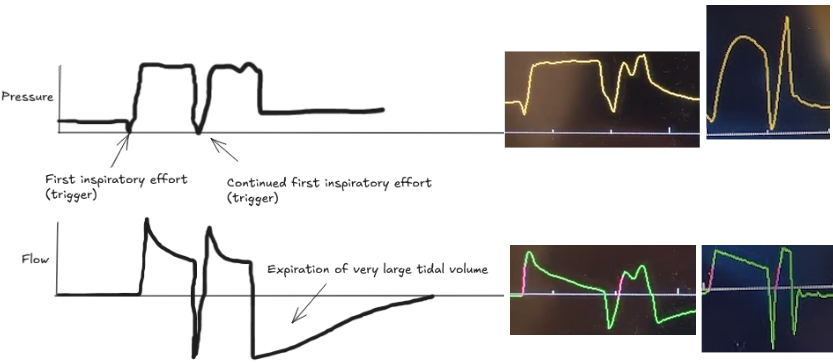
Troubleshooting options:
Dyssynchrony means the ventilator settings do not match the patient's needs. Watch the waveforms to diagnose the mismatch, then either adjust the ventilator or treat the underlying problem.
Thille, A. W., Rodriguez, P., Cabello, B., Lellouche, F., & Brochard, L. (2006). Patient-ventilator asynchrony during assisted mechanical ventilation. Intensive care medicine, 32, 1515-1522.
Blanch, L., Villagra, A., Sales, B., Montanya, J., Lucangelo, U., Luján, M., ... & Kacmarek, R. M. (2015). Asynchronies during mechanical ventilation are associated with mortality. Intensive care medicine, 41, 633-641.
Category: Critical Care
Keywords: ARDS (PubMed Search)
Posted: 6/16/2025 by Jordan Parker, MD
(Updated: 6/17/2025)
Click here to contact Jordan Parker, MD
Acute respiratory distress syndrome (ARDS) is an acute, inflammatory lung injury that effects the lung diffusely and can be triggered by various insults. Aside from the Kigali modification, the most recent updated definition of ARDS was the Berlin definition in 2012. There have been many advances and changes in the understanding and clinical practice for managing patients with ARDS since then. In 2024, Matthay, et al. proposed the new global definition to build upon the Berlin criteria [1]. They addressed several important issues with the Berlin definition to improve the diagnostic criteria and improve ability for diagnosis in resource-limited settings.
ARDS Berlin Definition
Important updates for the Global definition of ARDS
Diagnostic Criteria for the New Global Definition of ARDS from Matthay et al.
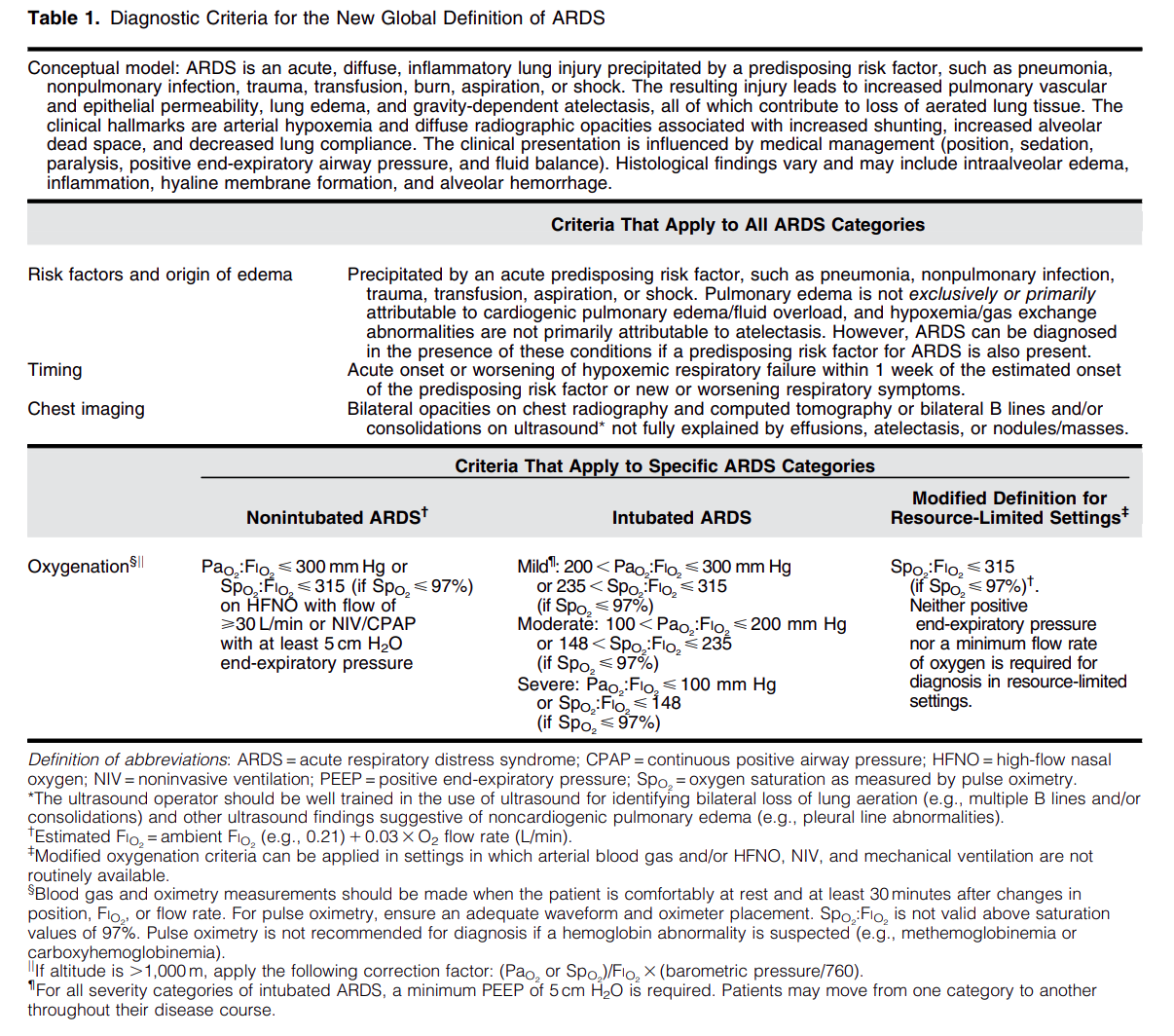
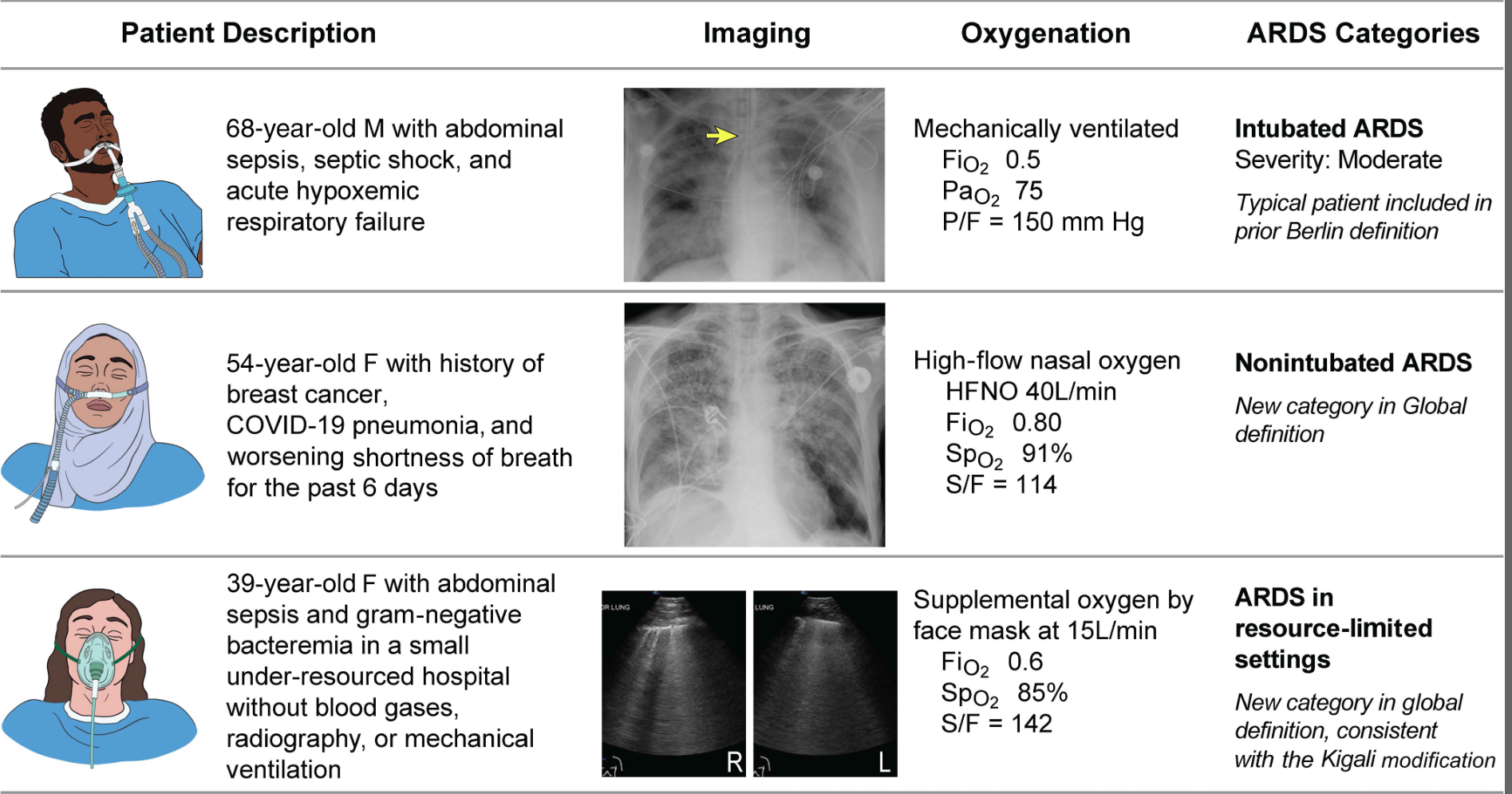
The Global Definition of ARDS expands upon the Berlin definition. It was shown that this new definition improves diagnosis in resource-limited settings, allows for earlier detection, and better classification [2]. A retrospective study evaluating this new global definition found that there was a significant number of patients identified using this new definition who would have been missed using the Berlin definition [3]. These patients may benefit from ARDS directed therapies and further prospective studies will be needed to assess how this new definition effects clinical management of these patients using the new definition.
Helpful table/figure from the paper.

Category: Critical Care
Keywords: Cardiac Arrest, PEA, ROSC (PubMed Search)
Posted: 6/9/2025 by Harry Flaster, MD
Click here to contact Harry Flaster, MD
Pulse Checks in Cardiac Arrest: Your Fingers Are Not Reliable.
Summary: Whenever possible, use an ultrasound or an arterial line for pulse checks. Our fingers are not reliable.
Key points:
Multiple studies have demonstrated that manual pulse checks are not a reliable method to determine ROSC. Arterial lines and ultrasound are far more reliable methods. However, using more accurate measures of circulation lead to an additional dilemma: at what MAP, SBP, or ultrasound measured flow should we stop chest compressions? There is no agreed upon number, and as with most dilemmas in clinical medicine, the best answer is, “it depends”. However, a MAP > 50 or SBP > 60 for most patients is a reasonable choice to stop chest compressions. MAP < 50 or SBP < 60 are unlikely to provide adequate perfusion to the brain, and chest compressions should be resumed.
References:
Category: Critical Care
Keywords: OHCA, shockable rhythms, VF, ventricular fibrillation, defibrillation, AED, energy (PubMed Search)
Posted: 6/4/2025 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
A recent retrospective cohort study out of China investigated an escalating energy (200 > 300 > 360J) versus fixed energy (200 > 200 > 200 J) defibrillation strategy in OHCA with ventricular fibrillation requiring repeated defibrillations.
Notes:
Results:
Caveats:
Category: Critical Care
Posted: 5/27/2025 by Quincy Tran, MD, PhD
(Updated: 5/13/2026)
Click here to contact Quincy Tran, MD, PhD
We have known that resuscitation with balanced crystalloids was associated with better outcomes, than normal saline. However, I have believed that in the early phase of resuscitation, volume of any crystalloids is still better than little volume. Thus, a couple of liters of normal saline (0.9% saline) would not hurt. However, the recent secondary analysis from the Crystalloid Liberal or Vasopressors Early Resuscitation in Sepsis (CLOVERS) trial might have changed my practice.
-----
Settings:
60 ICU in the United States between 2018 to 2022. This is the secondary analysis of the Crystalloid Liberal or Vasopressors Early Resuscitation in Sepsis (CLOVERS) trial population
Participants: Patients with sepsis-induced hypotension after receiving fluid resuscitation (from 1-3 litters). Participants who received lactate ringers (622 patients) were compared with patients who received normal saline (690 patients).
Outcome measurement:
Death before discharge home by day 90.
Hospital-free days at 28 days
Study Results:
Dead occurred in 12.2% of LR group (76/622) vs. 15.9% (110/690) patients of the NS group, Adjusted Hazard Ratio 0.71 (95% CI 0.51-0.99, p=0.043)
Patients receiving LR had more hospital-free days at 28 days than those receiving 0.9% saline (16.6 ± 10.8 vs. 15.4 ± 11.4 d, respectively). The mean difference was 1.6 days (95% CI, 0.4–2.8; p = 0.009).
Discussion:
This study confirms that not only early resuscitation is important, but the fluid choice during the early resuscitation phase is also important, especially in patients with signs and symptoms of sepsis.
Thus, even during pre-hospital phase and in the ED, clinicians should consider to use LR or other balanced solutions if available.
Conclusion:
Among patients with sepsis-induced hypotension, resuscitation with Lactate Ringer was associated with better outcomes than normal saline.
Gelbenegger G, Shapiro NI, Zeitlinger M, Jilma B, Douglas IS, Jorda A. Lactated Ringer's or Normal Saline for Initial Fluid Resuscitation in Sepsis-Induced Hypotension. Crit Care Med. 2025 May 1;53(5):e1140-e1144. doi: 10.1097/CCM.0000000000006601. Epub 2025 Feb 19. PMID: 39969246; PMCID: PMC12047640.
Category: Critical Care
Posted: 5/21/2025 by Caleb Chan, MD
(Updated: 5/13/2026)
Click here to contact Caleb Chan, MD
DeMasi et al. published a review on the current evidence surrounding peri-intubation and intubation practices. While the actual approach and context to each patient will be different it is good to be aware of the actual evidence base for medical decision-making.
Preoxygenation
Between Induction and Laryngoscopy
During Laryngoscopy and Intubation of the Trachea
Medications
Interventions to Prevent Hypotension
DeMasi SC, Casey JD, Semler MW. Evidence-based emergency tracheal intubation. Am J Respir Crit Care Med. Published online April 16, 2025.
Category: Critical Care
Posted: 5/15/2025 by William Teeter, MD
(Updated: 5/13/2026)
Click here to contact William Teeter, MD
Reversal of Factor Xa Inhibitor-Related Intracranial Hemorrhage: A Multicenter, Retrospective, Observational Study Comparing the Efficacy and Safety of Andexanet and PCCs
This study compared patients with a wide variety of intracranial hemorrhage types taking direct oral anticoagulants (e.g. apixaban or rivaroxaban) who then received andexanet versus prothrombin complex concentrates (PCCs)
Patients receiving andexanet (87.8%) had higher odds of achieving excellent/good hemostasis (odds ratio [OR] 1.60; 95% CI, 1.00-2.56; p = 0.048) compared with PCCs (81.8%). Patients treated with andexanet (7.9%) had higher odds of a thrombotic event (OR 1.91; 95% CI, 1.13-3.20; p = 0.014) compared to those treated with PCCs (4.2%).
This study found similar results to the previous ANNEXA-1 trial but included GCS < 7 or Neurosurgery within 12 hours of enrollment, which ANNEXA did not. This study was not designed to prove non-inferiority and should not change practice, especially given the eye-watering cost of Andexanet ($25,000+) versus PCCs (~$4000-6000)…. but betting they are working on one that will.
Category: Critical Care
Keywords: Noninvasive Ventilation, BiPAP, hypoxic respiratory failure (PubMed Search)
Posted: 5/6/2025 by Mark Sutherland, MD
(Updated: 5/13/2026)
Click here to contact Mark Sutherland, MD
Duan et al recently published in Intensive Care Medicine the results of a trial looking at a PEEP of 5 cm H2O vs 10 cm H2O and impact on failure rate (progression to intubation) when using non-invasive ventilation (NIV). In their trial, the high PEEP group had a lower rate of intubation (32% vs 43%), and this was statistically significant. It is important to note that they excluded patients whose indication for NIV was heart failure, asthma, or COPD exacerbation.
Ultimately, how to choose the right PEEP is a very complex question and requires tailoring to your patient's physiology and clinical circumstances. For example, hypercarbic patients may benefit more from a maximization of their driving pressure (Pplat - PEEP), which can involve lowering their PEEPs, especially when trying to avoid gastric insufflation (remember, pressures of 30 cm of H2O or higher are very likely to open the LES).
Bottom Line: PEEP and other vent settings should be tailored to the patient's pathophysiology, but this trial suggests that in hypoxemic patients not getting NIV for heart failure, asthma, or COPD exacerbation, a higher PEEP (10 vs 5) may reduce the risk of intubation.
Duan, J., Liu, X., Shu, W. et al. Low versus high positive end expiratory pressure in noninvasive ventilation for hypoxemic respiratory failure: a multicenter randomized controlled trial. Intensive Care Med (2025). https://doi-org.proxy-hs.researchport.umd.edu/10.1007/s00134-025-07902-4
Category: Critical Care
Posted: 4/29/2025 by Mike Winters, MBA, MD
(Updated: 5/13/2026)
Click here to contact Mike Winters, MBA, MD
Sedation for the Mechanically Ventilated Adult ICU Patient
Lewis K, et al. Executive summary of a focused update to the clinical practice guidelines for the prevention and management of pain, anxiety, agitation/sedation, delirium, immobility, and sleep disruption in adult patients in the ICU. Crit Care Med. 2025; 53:e701-e710.
Category: Critical Care
Posted: 4/22/2025 by Cody Couperus-Mashewske, MD
Click here to contact Cody Couperus-Mashewske, MD
This large RCT compared High-Flow Nasal Oxygen (HFNO) against Noninvasive Ventilation (NIV) via face mask in 5 types of Acute Respiratory Failure (ARF): non-immunocompromised hypoxemia, immunocompromised hypoxemia, COPD with acidosis, acute cardiogenic pulmonary edema (ACPE), and COVID-19.
Bottom Line:
RENOVATE suggests HFNO might be a reasonable, more comfortable initial choice for non-immunocompromised hypoxemic ARF or COVID-19 ARF. However, exercise caution using HFNO first-line for COPD exacerbations with acidosis or immunocompromised hypoxemic ARF due to conflicting analyses and potential harm signals. The signal for HFNO benefit in ACPE is intriguing but needs confirmation before changing practice. Close monitoring for failure and timely escalation are essential regardless of the initial noninvasive strategy.
High-flow nasal oxygen vs noninvasive ventilation in patients with acute respiratory failure: the RENOVATE randomized clinical trial RENOVATE Investigators and the BRICNet Authors, Maia IS, Kawano-Dourado L, et al. JAMA. Published online December 10, 2024. doi:10.1001/jama.2024.26244
Category: Critical Care
Posted: 3/22/2025 by Jordan Parker, MD
(Updated: 4/15/2025)
Click here to contact Jordan Parker, MD
Background:
Acetaminophen can reduce hemoprotein induced oxidative damage. There has been growing discussion about its benefits in critically ill patients with sepsis. Multiple observational studies have found conflicting results on mortality in critically ill patients with sepsis. The ASTER trial found no difference in number of days alive and free of organ support. Interestingly their secondary outcomes found significantly less development of ARDS in the acetaminophen group 2.2% vs 8.5%, p = .01. There was also a non-statistically significant difference in mortality between the groups in favor of the acetaminophen group, 17% vs 22% p = 0.19. This study looked to further evaluate if acetaminophen used in critically ill patients with sepsis would have a decrease in mortality and increase in ventilator free days.
Study:
- Retrospective analysis of the Ibuprofen in Sepsis Study (ISS)
- The ISS was a randomized clinical trial comparing ibuprofen with placebo in critically ill patients with sepsis. Careful documentation of Acetaminophen use was recorded for the trial
- Critically-ill adults across 7 ICU’s in the US and Canada with known or suspected infection and severe organ dysfunction
- Acetaminophen use within 48 hours of enrollment = Acetaminophen exposed
- Primary outcome: 30-day mortality
- Secondary outcome: Renal failure and ventilator free days up to day 28
- 455 patients. 172 Acetaminophen unexposed, 235 Acetaminophen exposed.
Results:
- Propensity-matched analysis showed a lower mortality risk at 30 days in patients exposed to acetaminophen compared to unexposed, 32% vs 49% (HR 0.58, p .004)
- Secondary outcomes found acetaminophen exposed group had more ventilator free days (p .02) but there was no difference in renal failure among the groups.
Limitations:
- Major risk for confounding variables
- Retrospective and the data used was from decades ago (1989 -1995). Sepsis care has evolved and improved since this time
- Dose and frequency of acetaminophen administration was not standardized
Take Home Points:
- Interesting research that provides further support on the possible benefit to using acetaminophen in the management of critically ill patients with sepsis.
- With the ASTER trial showing a signal for the decrease in development of ARDS and this study showing improvement in mortality one could make a case for starting acetaminophen early in the course for these patients. However, the data is conflicting and more prospective, RCT’s are needed to confirm these findings before making this a staple for sepsis care in critically ill patients.
Obeidalla, S. N., Bernard, G. R., Ware, L. B., & Kerchberger, V. E. Acetaminophen and Clinical Outcomes in Sepsis: A Retrospective Propensity Score Analysis of the Ibuprofen in Sepsis Study. CHEST Critical Care. 2025;3(1):100-118. https://doi.org/10.1016/j.chstcc.2024.100118
Ware LB, Files DC, Fowler A, et al. Acetaminophen for Prevention and Treatment of Organ Dysfunction in Critically Ill Patients With Sepsis: The ASTER Randomized Clinical Trial. JAMA. 2024;332(5):390–400. doi:10.1001/jama.2024.8772
Category: Critical Care
Keywords: Catheter, flow rates, resuscitation (PubMed Search)
Posted: 4/9/2025 by Harry Flaster, MD
Click here to contact Harry Flaster, MD
Flow rates are, in theory, determined by Poiseuille’s Law, which states that the flow rate depends on fluid viscosity, pipe length, and the pressure difference between the ends of the pipe .
Of course we won’t be calculating this during a resuscitation! Nor would it be useful if we did: the equation assumes laminar flow, whereas turbulent flow is more likely. Nor is it practical to look up the viscosity of crystalloid/blood/plasma, which also dramatically impacts flow rates.
Instead, remember this equation: Larger + shorter = faster
And keep in mind the following:
In practice, our friends in Australia actually put common catheters to the test, and provided these helpful results:
Or, as a picture:
Note, these flow rates were achieved using crystalloid. Blood will be slower due to higher viscosity.
Category: Critical Care
Keywords: OHCA, cardiac arrest, refractory VT/VF, shockable, ventricular arrhythmia, amiodarone, lidocaine (PubMed Search)
Posted: 4/2/2025 by Kami Windsor, MD
(Updated: 5/13/2026)
Click here to contact Kami Windsor, MD
A 2023 retrospective cohort study comparing amiodarone to lidocaine for in-hospital cardiac arrests (IHCA) with refractory VT/VF found that use of lidocaine was associated with increased chance of ROSC, 24 hour survival, survival to discharge, and favorable neurologic outcome at hospital discharge.[1]
Now, a recent study comparing amiodarone to lidocaine in the pre-hospital setting for OHCA has found similar results. [2] Another retrospective cohort study using propensity score matching, they evaluated 23,263 adult patients with OHCA and defibrillation refractory VT/VF managed by 1700 EMS agencies.
Use of lidocaine was associated with greater odds of prehospital ROSC, fewer post-drug administration defibrillations, and greater odds of survival to discharge.
In comparison to earlier trials, these studies are some of the first demonstrating benefits to lidocaine use over amiodarone that reach statistical significance, but of course have all the limitations that come with retrospective studies and are not further analyzed in the context of etiologies for cardiac arrest or application of post-ROSC care.
Bottom Line: If you happen to be someone who reaches for amiodarone as your go-to, it may be time to start considering lidocaine.
Category: Critical Care
Keywords: community acquired pneumonia; CAP; corticosteroids; mortality; adjuvant therapy (PubMed Search)
Posted: 3/25/2025 by Quincy Tran, MD, PhD
Click here to contact Quincy Tran, MD, PhD
If you watch those medical drama (House MD, ER, Grey’s Anatomy, Resident…), the doctors and residents are always faced with a dilemma – is it a rare autoimmune disorder or is it an infection? They are worried that if they give steroid to a patient with infections, that would kill the patients.
Well, it might not be the case for Community acquired pneumonia.
A meta-analysis of randomized control trials involving 3224 patients to look into the efficacy of adjuvant corticosteroids for CAP. The authors assessed the heterogeneity of treatment effect (different groups should have different response to treatment).
For patients who were anticipated to benefit (those who had CRP > 240 mg/L), corticosteroids were associated with lower odds of 30-day mortality (OR 0·43 [0·25–0·76], p=0·026).
When stratifying by risk, there was no significant effect between those with Pneumonia Severity Index (PSI) I-III versus those with PSI IV-V.
However, corticosteroids increased odds of hyperglycemia (OR 2·50 [95% CI 1·63–3·83], p<0·0001), odds of hospital readmissions (1·95 [1·24–3·07], p=0·0038)
Discussion:
There were different regiments for corticosteroids in the included studies. However, hydrocortisone appeared to be more effective than other corticosteroids.
Furthermore, the time intervals for treatment is still debatable. The data suggested that the ideal treatment is within 24 hours of hospital admission, but patients can still benefit from treatment in up to 48 hours.
A response-dependent treatment is also recommended: 8 days or 14 days, depending on how patients respond to treatment by day 4.
Conclusion:
Adjuvant treatment with corticosteroids among hospitalized patients with CAP was significantly associated with reduction of 30-day mortality. The treatment effect, however, varied according to patients CRP concentrations at baseline.
Smit JM, Van Der Zee PA, Stoof SCM, Van Genderen ME, Snijders D, Boersma WG, Confalonieri P, Salton F, Confalonieri M, Shih MC, Meduri GU, Dequin PF, Le Gouge A, Lloyd M, Karunajeewa H, Bartminski G, Fernández-Serrano S, Suárez-Cuartín G, van Klaveren D, Briel M, Schönenberger CM, Steyerberg EW, Gommers DAMPJ, Bax HI, Bos WJW, van de Garde EMW, Wittermans E, Grutters JC, Blum CA, Christ-Crain M, Torres A, Motos A, Reinders MJT, Van Bommel J, Krijthe JH, Endeman H. Predicting benefit from adjuvant therapy with corticosteroids in community-acquired pneumonia: a data-driven analysis of randomised trials. Lancet Respir Med. 2025 Mar;13(3):221-233. doi: 10.1016/S2213-2600(24)00405-3. Epub 2025 Jan 29. PMID: 39892408.
Category: Critical Care
Keywords: Mechanical Ventilation, Brain Injury, ICH, Stroke, Hypercapnea, Hypoxia, Hyperoxia (PubMed Search)
Posted: 3/4/2025 by Mark Sutherland, MD
Click here to contact Mark Sutherland, MD
Intubation and mechanical ventilation of brain injured patients, which is extremely common in the Emergency Department, can be very challenging and subject to significant practice variation. It is often said that brain injured patients “can't take a joke”, meaning that they are less tolerant to hemodynamic and metabolic perturbations, and these perturbations tend to be associated with very large swings in their clinical outcomes. For example, hypo/hyperglycemia, hypo/hypernatremia, hypo/hypertension, hypo/hyperoxia, hypo/hypercapnea, etc are all extremely important to avoid. This is probably the one patient population where “euboxia” (the notion that we obsess too much about making all the numbers pretty in the EMR) is probably not as applicable. As such, there is at least good physiologic rationale, and now increasing empirical evidence, that ventilating these patients very thoughtfully is extremely important and likely to have meaningful impact on patient-oriented outcomes (mortality, neurologic outcome, etc).
The VENTIBRAIN study was a prospective observation trial of 2,095 intubated patients in 26 countries who had TBI, ICH (including SAH), or acute ischemic stroke. Interestingly, they found that patients with lower tidal volume (TV) per predicted body weight had higher mortality (although the majority of their TVs were well controlled and in a fairly tight range), which is contrary to conventional thinking in pulmonary pathologies like ARDS. They also found that higher driving pressure (DP) was associated with higher mortality, which agrees with data from other conditions. PEEP and FiO2 had U-shaped curves, but FiO2 in particular tended to favor lower FIO2, also similar to current thinking for ICU patients in general.
Take Home Points:
Learning Driving Pressure/PEEP Titration:
