Category: Critical Care
Keywords: OHCA, cardiac arrest, resuscitation, PEA, pesudo-PEA, pulseless electrical activity (PubMed Search)
Posted: 11/12/2019 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
When managing cardiac arrest, it is important to differentiate PEA, the presence of organized electrical activity without a pulse, from "pseudo-PEA,"where there is no pulse but there IS cardiac activity visualized on ultrasound.
Why:
How:
What:
Bottom Line: Pseudo-PEA is different from PEA. Utilize POCUS during your cardiac arrests to identify it and to help diagnose reversible causes, and treat it as a profound shock state with the appropriate supportive measures, i.e. pressors or inotropy.
Rabjohns J, Quan T, Boniface K, Pourmand A. Pseudo-pulseless electrical activity in the emergency department, an evidence based approach. Am J Emerg Med. 2019. DOI:https://doi.org/10.1016/j.ajem.2019.158503
Category: Critical Care
Keywords: VAPI, acute respiratory failure, vaping, e-cigarettes, e-hookah, juul, pulmonary disease, acute lung diease, ARDS (PubMed Search)
Posted: 9/23/2019 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
The U.S. is currently experiencing an epidemic of a severe lung disease termed Vaping-Associated Pulmonary Illness (VAPI), with over 500 cases and 7 deaths across 38 states and 1 U.S. territory since July 2019.
The clinical presentation of VAPI varies --
Diagnostics --
Treatment is supportive +/- steroids --
Bottom Line: Include vaping-associated pulmonary illness in your differential for patients presenting with acute lung disease.
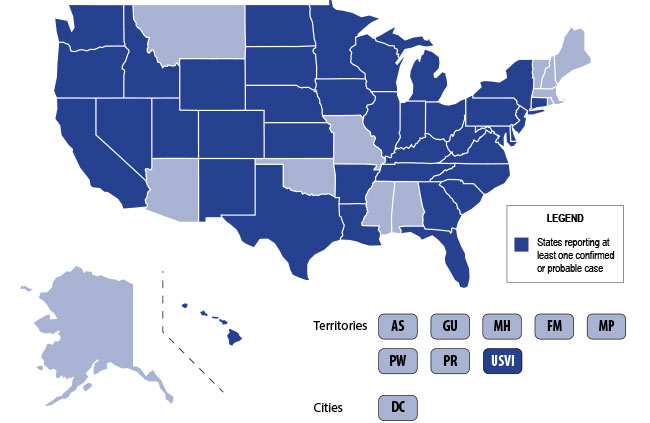
Background: The use of electronic nicotine delivery systems, also known as e-cigarettes or vape pens, has risen precipitously since their introduction in 2006. They heat a liquid that can contain nicotine, THC/CBD, flavors and/or other additives, producing an aersol that is inhaled by users.
They have been marketed as a way to quit smoking, and as being safer than cigarettes. The U.S. is, however, currently experiencing an epidemic of a severe lung disesae termed Vaping-Associated Pulmonary Illness, with over 500 cases and 7 deaths across 38 states and 1 U.S. territory.
Per data obtained by the CDC:
CDC Emergency Preparedness & Response: "Severe Pulmonary Disease Associated with Using E-Cigarette Products" https://emergency.cdc.gov/han/han00421.asp
Category: Critical Care
Keywords: mechanical ventilation, respiratory failure, obstructive lung disease, asthma exacerbation, COPD (PubMed Search)
Posted: 8/6/2019 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
Managing the intubated patient with exacerbation of severe obstructive lung disease, especially asthma, can be very challenging as it carries higher risks of barotrauma due to higher pulmonary pressures and circulatory collapse due to auto-PEEP and decreased venous return. When measures such as medical therapy and noninvasive positive-pressure ventilation fail to prevent intubation, here are some tips to help:
1. Utilize a volume control ventilation mode to ensure a set tidal volume delivery / minute ventilation, as pressure-targeted modes will be more difficult due to the high pulmonary pressures in acute obstructive lung disease.
2. Set a low RR in order to allow for full exhalation, avoiding air-trapping / breath-stacking and circulatory collapse due to decreased venous return. This may require deep sedation and potentially paralysis.
3. Increase your inspiratory flow by shortening your inspiratory time (thereby increasing your time for exhalation.
4. Monitor for auto-PEEP:
5. Peak inspiratory pressures will be high -- what is more important is the plateau pressure, measured by performing an inspiratory hold at the end of inspiration. Provided your plateau pressure remains <30, you don't need to worry as much about the peak pressure alarms.
6. If your patient acutely decompensates in terms of hemodynamics and oxygenation -- first attempt to decompress their likely auto-PEEPed lungs by popping them off the ventilator and manually press on their chest to assist with exhalation of stacked breaths allowing venous return to the heart.
Category: Critical Care
Keywords: Resuscitation, cardiac arrest, POCUS, ultrasound, ROSC (PubMed Search)
Posted: 4/9/2019 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
Background: Previous systematic reviews1,2,3 have indicated that the absence of cardiac activity on point-of-care ultrasound (POCUS) during cardiac arrest confers a low likelihood of return of spontaneous circulation (ROSC), but included heterogenous populations (both traumatic and atraumatic cardiac arrest, shockable and nonshockable rhythms).
The SHoC investigators4 are the first to publish their review of nontraumatic cardiac arrests with nonshockable rhythms, evaluating POCUS as predictor of ROSC, survival to admission (SHA), and survival to discharge (SHD) in cardiac arrests occurring out-of-hospital or in the ED.
Bottom Line: In nontraumatic cardiac arrest with non-shockable rhythms, the absence of cardiac activity on POCUS may not, on its own, be as strong an indicator of poor outcome as previously thought.
Category: Critical Care
Keywords: heart transplant, arrhythmias, critical care (PubMed Search)
Posted: 3/26/2019 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
When managing transplant patients it is important to keep in mind the anatomic and physiologic changes that occur with the complete extraction of one person's body part to replace another's.
For cardiac transplant patients with symptomatic bradycardia:
For cardiac transplant patients with tachyarrythmias:
Stecker EC, Strelich KR, Chugh SS, et al. Arrythmias after orthotopic heart transplantation. J Card Fail. 2005;11(6):464-72.
Thajudeen A, Stecker EC, Shehata M, et al. Arrhythmias after heart transplantation: Mechanisms and management. J Am Heart Assoc. 2012;1(2):e001461.
Category: Critical Care
Keywords: Airway management, acute respiratory failure, hypoxia, intubation, preoxygenation (PubMed Search)
Posted: 3/12/2019 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
The PROTRACH study recently compared preoxygenation with standard bag valve mask (BVM) at 15 lpm to preoxygenation + apneic oxygenation with high flow nasal cannula 60 lpm/100% FiO2 in patients undergoing rapid sequence intubation.
Guitton C, Ehrmann S, Volteau C, et al. Nasal high-flow preoxygenation for endotracheal intubation in the critically ill patient: a randomized clinical trial. Intensive Care Med. 2019. doi: 10.1007/s00134-019-05529-w. [Epub ahead of print]
Category: Critical Care
Keywords: ARDS, respiratory failure, ventilator settings, critical care (PubMed Search)
Posted: 2/26/2019 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
Despite ongoing research and efforts to improve our care of patients with ARDS, it remains an entity with high morbidity and mortality. Early recognition of the disease process and appropriate management by emergency physicians can have profound effects on the patient's course, especially in centers where ICU boarding continues to be an issue.
Recognition of ARDS (Berlin criteria)
*An ABG should be obtained in the ED if physicians are unable to wean down FiO2 from high settings, if oxygenation by pulse ox is marginal, or if the patient is in a shock state.
Tenets of ARDS Management:
*IBW Males = 50 + 2.3 x [Height (in) - 60] / IBW Females = 45.5 + 2.3 x [Height (in) - 60]
Strategies for Refractory Hypoxemia in the ED: You can't prone the patient, but what else can you do?
1. Escalate PEEP in stepwise fashion
2. Recruitment maneuvers
3. Appropriate sedation and neuromuscular blockade
4. Inhaled pulmonary vasodilators (inhaled prostaglandins, nitric oxide) if known or suspected right heart failure or pulmonary hypertension
Bottom Line: Emergency physicians are the first line of defense against ARDS. Early recognition of the disease process and appropriate management is important to improve outcomes AND to help ICU physicians triage which patients need to be emergently proned or even who should potentially be referred for ECMO.
Fielding-Singh V, Matthay MA, Calfee CS. Beyond Low Tidal Volume Ventilation: Treatment Adjuncts for Severe Respiratory Failure in Acute Respiratory Distress Syndrome. Crit Care Me.. 2018;46(11):1820-31.
Category: Critical Care
Keywords: neutropenic fever, typhlitis, necrotizing enterocolitis, sepsis, septic shock (PubMed Search)
Posted: 2/12/2019 by Kami Windsor, MD
(Updated: 5/13/2026)
Click here to contact Kami Windsor, MD
Neutropenic enterocolitis can occur in immunosuppressed patients, classically those being treated for malignancy (hematologic much more commonly than solid tumor). When involving the cecum specifically, it is known as "typhlitis."
It should be considered in any febrile neutropenic patients with abdominal pain or other symptoms of GI discomfort (diarrhea, vomiting, lower GI bleeding), and can be confirmed with CT imaging.
A recent study found that invasive fungal disease, most often candidemia, occurred in 20% of febrile neutropenic patients with CT-confirmed enteritis, a rate that increased to 30% if the patient was in septic shock.
Take Home:
1. Have a lower threshold for abdominal CT imaging in your patients with febrile neutropenia and abdominal pain/GI symptoms, especially if they are critically ill.
2. Consider addition of IV antifungal therapy if they are hemodynamically unstable with enterocolitis on CT.
Duceau B, Picard M, Pirrachio R, et al. Neutropenic enterocolitis in critically ill patients: Spectrum of the disease and risk of invasive fungal disease. Crit Care Med. 2019. [Epub ahead of print] doi: 10.1097/CCM.0000000000003687.
Category: Critical Care
Keywords: OHCA, cardiac arrest, resuscitation, maternal cardiac arrest, pregnancy (PubMed Search)
Posted: 1/29/2019 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
Historically, there has been very limited data regarding the epidemiology of OHCA in pregnant females. Two recently-published studies tried to shed some light on the issue.
Both Maurin et al.1 and Lipowicz et al.2 looked at all-cause out-of-hospital maternal cardiac arrest (MCA) data in terms of numbers and management, in Paris and Toronto respectively, from 2009/2010 to 2014. Collectively, they found:
A few reminders from the 2015 AHA guidelines for the management of cardiac arrest in pregnancy:
Bottom Line: Although maternal cardiac arrest is relatively rare, survival in OHCA is lower than perhaps previously thought. Areas to improve include public education on the importance of bystander CPR in pregnant females, and appropriate physician adherence to PMCS recommendations, with decreased on-scene time by EMS in order to decrease time to PMCS.
Maurin et al. looked at documented out-of-hospital maternal cardiac arrest (MCA) in pregnant females ≥18 years old, in Paris from 2009 to 2014 and reported on some aspects of prehospital care. Prehospital management there includes activation of both a BLS (which usually arrives first) and ALS team, with a prehospital emergency physician being a member of the ALS team.
Lipowicz et al. similarly looked at MCA from 2010 to 2014 using data from the Toronto Regional RescuNet cardiac arrest database:
Category: Critical Care
Keywords: circulatory dysfunction, hypotension, shock, fluid resuscitation, IV fluids (PubMed Search)
Posted: 1/1/2019 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
The European Society of Intensive Care Medicine (ESICM) recently released a review with recommendations from an expert panel for the use of IV fluids in the resuscitation of patients with acute circulatory dysfunction, especially in settings where invasive monitoring methods and ultrasound may not be available.
Points made by the panel include:
Recommendations from the panel include:
Bottom Line: Utilize all the information you have about your patient to determine whether or not they require IVF, and reevaluate their physical and biochemical (lactate) response to fluids to ensure appropriate IVF administration and avoid volume overload.
Cecconi M, Hernandez G, Dunser M, et al. Intensive Care Med. 2018. https://doi-org.proxy-hs.researchport.umd.edu/10.1007/s00134-018-5415-2
Category: Critical Care
Keywords: hyperoxia, oxygen therapy, saturation, SpO2, critical care, mechanical ventilation (PubMed Search)
Posted: 12/4/2018 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
Hyperoxia has been repeatedly demonstrated to be detrimental in a variety of patients, including those with myocardial infarction, cardiac arrest, stroke, traumatic brain injury, and requiring mechanical ventilation,1-4 and the data that hyperoxia is harmful continues to mount:
Bottom Line: Avoid hyperoxia in your ED patients, both relatively stable and critically ill. Remove or turn down supplemental O2 added by well-meaning pre-hospital providers and nurses, and wean down ventilator settings (often FiO2). A target SpO2 of >92% (>88% in COPD patients) or PaO2 >55-60 is reasonable in the majority of patients.8
Category: Critical Care
Keywords: resuscitation, liver failure, cirrhosis (PubMed Search)
Posted: 11/20/2018 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
A few (out of 10) tips for the care of sick patients with liver failure:
Fuhrmann V, Whitehouse T, Wendon J. The ten tips to manage critically ill patients with acute-on-chronic liver failure. Intensive Care Med. 2018;44(11):1932-5.
Category: Critical Care
Keywords: resuscitation, cardiac arrest, post-cardiac arrest care, blood pressure, MAP, ROSC (PubMed Search)
Posted: 11/5/2018 by Kami Windsor, MD
(Updated: 11/6/2018)
Click here to contact Kami Windsor, MD
The most recent AHA guidelines for goal blood pressure after return of spontaneous circulation (ROSC) post-cardiac arrest recommend a definite mean arterial pressure (MAP) goal of > 65 mmHg.1 There is no definitive data to recommend a higher specific goal, but there is some evidence to indicate that maintaining higher MAPs may be associated with better neurologic outcomes.2
A recently published prospective, observational, multicenter cohort study looked at neurologic outcomes corresponding to different MAPs maintained in the initial 6 hours post-cardiac arrest.3
Findings:
1. Compared to lower blood pressures (MAPs 70-90 mmHg), the cohort with MAPs > 90 mmHg had:
2. The association between MAP > 90 mmHg and good neurologic outcome was stronger among patients with a previous diagnosis of hypertension, and persisted regardless of initial rhythm, use of vasopressors, or whether the cardiac arrest occured in or out of hospital.
3. There was a dose-response increase in probability of good neurologic outcome among all MAP ranges above 90 mmHg, with MAP >110 mmHg having the strongest association with good neurologic outcome at hospital discharge.
Note: The results of a separate trial, the Neuroprotect post-CA trial, comparing MAPs 85-100 mmHg to the currently recommended MAP goal of >65 mmHg, are pending.4
Bottom Line: As per current AHA guidelines, actively avoid hypotension, and consider use of vasopressor if needed to maintain MAPs > 90 mmHg in your comatose patients post-cardiac arrest, especially those with a preexisting diagnosis of hypertension.
Category: Critical Care
Keywords: High flow nasal cannula, acute respiratory failure, hypoxia, hypercarbia, non-invasive ventilation (PubMed Search)
Posted: 10/9/2018 by Kami Windsor, MD
(Updated: 5/13/2026)
Click here to contact Kami Windsor, MD
We know that high flow nasal cannula is an option in the management of acute hypoxic respiratory failure without hypercapnea. A newer iteration of high flow, "high velocity nasal insufflation" (HVNI), may be up-and-coming.
According to its makers (Vapotherm), it is reported to work mainly by using smaller bore nasal cannulae that deliver the same flows at higher velocities, thereby more rapidly and repeatedly clearing dead space, facilitating gas exchange and potentially offering ventilatory support.
In an industry-sponsored non-inferiority study published earlier this year:
Bottom Line:
The availability of a nasal cannula that helps with CO2 clearance would be great, and an option for patients who can't tolerate the face-mask of NPPV would be even better.
HVNI requires more investigation with better studies and external validation before it can really be considered noninferior to NPPV, but it certainly is interesting.
Category: Critical Care
Keywords: acidosis, acidemia, sodium bicarbonate, shock (PubMed Search)
Posted: 9/11/2018 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
The recently published BICAR-ICU study looked at the use of bicarb in critically ill patients with severe metabolic acidemia...
Bottom Line:
Consider administration of sodium bicarbonate for your critically ill ED patients with severe metabolic acidosis and AKI, especially if acidosis &/or renal function is not improved with usual initial measures (such as IVF, etc).
*Acute Kidney Injury Network Staging Criteria

Jaber S, Paugam C, Futier E, et al. Sodium bicarbonate therapy for patients with severe metabolic acidaemia in the intensive care unit (BICAR-ICU): a multicentre, open-label, randomised controlled, phase 3 trial. Lancet. 2018;392(10141):31-40.
Category: Critical Care
Keywords: Resuscitation, OHCA, prehospital medicine, cardiac arrest, epinephrine (PubMed Search)
Posted: 8/14/2018 by Kami Windsor, MD
(Updated: 5/13/2026)
Click here to contact Kami Windsor, MD
The highly-awaited PARAMEDIC2 trial results are in:
Interestingly, the authors also queried the public as to what mattered to them most:

Bottom Line:
A Few Things:
Perkins GD, Ji C, Deakin CD, et al. A Randomized Trial of Epinephrine in Out-of-Hospital Cardiac Arrest. N Engl J Med. 2018. doi: 10.1056/NEJMoa1806842.
Category: Critical Care
Keywords: noninvasive positive pressure ventilation, NIV, NIPPV, DNI, do-not-intubate, palliative care, end-of-life, respiratory distress (PubMed Search)
Posted: 7/17/2018 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
When a do-not-intubate (DNI) hospice patient arrives in the ED with respiratory distress, consideration of non-invasive positive pressure ventilation (NIPPV) could invoke either a “What other option do I have?” or “Why torture the patient and prolong the dying process?” sentiment.
But what’s the data?
A recently-published meta-analysis1 found that in DNI patients receiving NIPPV, there was a 56% survival rate to hospital discharge and 32% survival to 1-year.
Independent studies have demonstrated:
Bottom Line:
Category: Critical Care
Keywords: cardiac arrest, CPR, obesity (PubMed Search)
Posted: 6/19/2018 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
Although not specifically a part of current recommendations due to lack of data, the AHA has previously recommended shifting upward on the sternum during CPR in the pulseless pregnant patient in order to account for upward displacement of the heart by a gravid uterus. Should the same be done for our obese patients?
Lee et al. performed a retrospective study that reviewed chest CTs to determine the location on the sternum that corresponded to the optimal point of maximal left ventricular diameter (OPLV), in both obese and non-obese patients.
They found that the OPLV was higher (more cranial) on the sternum for obese patients than for patients with normal weight, although 96% of obese patients' OPLV fell within 2cm of where the guidelines recommend standard hand placement should be, compared to a notable 52% in non-obese patients.

*as measured from the distal end of the sternum
Bottom Line: Radiographically, the location on the sternum that corresponds to optimal compression of the LV is more cranial in obese patients than in non-obese patients. It remains to be seen whether the recommendations for hand placement in CPR should be adjusted, but we may want to consider staying within 4cm of the bottom of the sternum in patients of normal weight.
Lee J, Oh J, Lim TH, et al. Comparison of optimal point on the sternum for chest compression between obese and normal weight individuals with respect to body mass index, using computer tomography: A retrospective study. Resuscitation. 2018; 128:1-5.
Category: Critical Care
Keywords: sepsis, septic shock, guidelines (PubMed Search)
Posted: 5/22/2018 by Kami Windsor, MD
Click here to contact Kami Windsor, MD


Take Home Points:
For additional reading:
EMNerd, Dr. Rory Spiegel https://emcrit.org/emnerd/em-nerd-case-temporal-fallacy/
Surviving Sepsis Campaign http://www.survivingsepsis.org/Guidelines/Pages/default.aspx
Category: Critical Care
Keywords: acute hypoxic respiratory failure, intubation, noninvasive positive pressure ventilation, high flow nasal cannula, BiPAP, vapotherm (PubMed Search)
Posted: 4/25/2018 by Kami Windsor, MD
Click here to contact Kami Windsor, MD
ED physicians frequently utilize modailities such as noninvasive positive pressure ventilation (NIV) and high flow nasal cannula (HFNC) to support and potentially avoid intubation in patients presenting with acute hypoxic respiratory failure. Unfortunately, failure of these measures, resulting in "delayed" intubation, has been associated with increased mortality.1,2
A recent post-hoc analysis of data from a multicenter randomized controlled trial evaluated 310 patients with acute hypoxic respiratory failure managed with supplemental O2 by regular nasal cannula, HFNC, or NIV.3
The following factors were predictive of eventual intubation in the different groups:
Of note, 45% of the 310 patients eventually required intubation, and these patients in general had a higher initial respiratory rate and lower PaO2 at presentation, and were more likely to have bilateral infiltrates on CXR.
Bottom Line: Reevaluate your patients frequently. If RR remains high, P:F ratio remains low, or patient respiratory effort/work of breathing is not alleviated by noninvasive measures, consider pulling the trigger on intubation earlier.
