Category: Critical Care
Keywords: thoracentesis, pleural effusion, critical care (PubMed Search)
Posted: 5/4/2015 by Feras Khan, MD
Click here to contact Feras Khan, MD
Safety of Thoracentesis
Results after 24 hours of followup post-procedure
Other interesting points:
Bottom line: Use your ultrasound to direct your tap and dont take out more than 1500 mL routinely
Ault MJ et al. Thoracentesis outcomes: a 12-year experience. Thorax 2015;70:127-132.
Category: Critical Care
Posted: 4/28/2015 by Mike Winters, MBA, MD
Click here to contact Mike Winters, MBA, MD
SIRS and Severe Sepsis Screening
Kaukonen KM, Bailey M, Pilcher D, et al. Systemic Inflammatory Response Syndrome Criteria in Defining Severe Sepsis. NEJM 2015;372:1629-38.
Category: Critical Care
Keywords: large hemispheric infarct, acute ischemic infarct, stroke (PubMed Search)
Posted: 4/20/2015 by John Greenwood, MD
(Updated: 4/21/2015)
Click here to contact John Greenwood, MD
Updates in the Management of Large Hemispheric Infarction
Large hemispheric infarctions (LHI) are estimated to occur in 2-8% of all hospitalized ischemic strokes and 10 15% of all MCA territory infarcts. LHI carry high rates of morbidity and mortality, in fact, if left untreated associated cerebral edema can rapidly progress to transtentorial herniation and death in 40 80% of patients.
Recognized risk factors for progressive cerebral edema include:
Evidence based medical strategies for LHI include:
Prophylactic hemicraniectomy
Bottom Line: Early recognition of large hemispheric stroke is critical as it is associated with a high rate of morbidity and mortality. Aggressive medical management and early neurosurgical involvement may improve outcomes.
References
Follow me on Twitter @JohnGreenwoodMD
Category: Critical Care
Posted: 4/14/2015 by Haney Mallemat, MD
Click here to contact Haney Mallemat, MD
You decide to do a R.U.S.H. exam on your hypotensive patient and perform an apical four-chamber view.You see one of the two clips below; are there any tricks to figure out which is the left ventricle and which is the right ventricle?
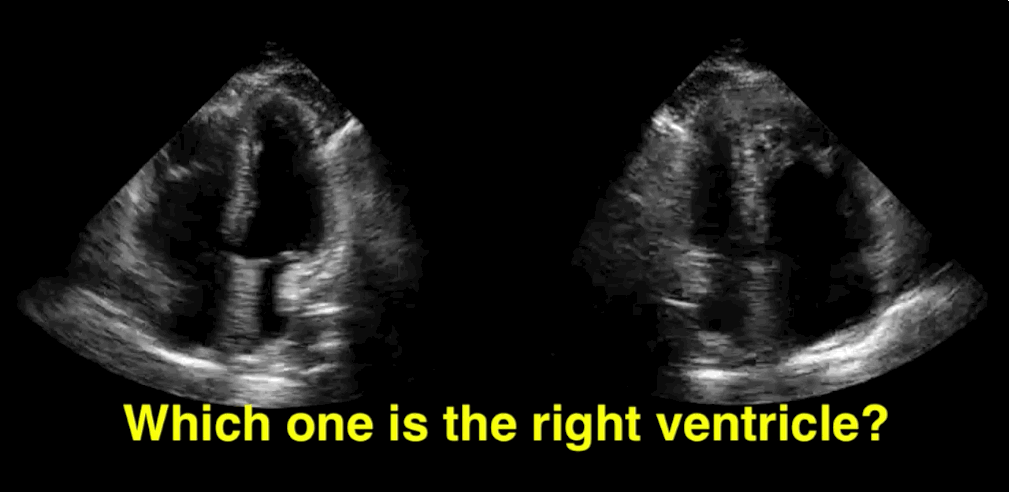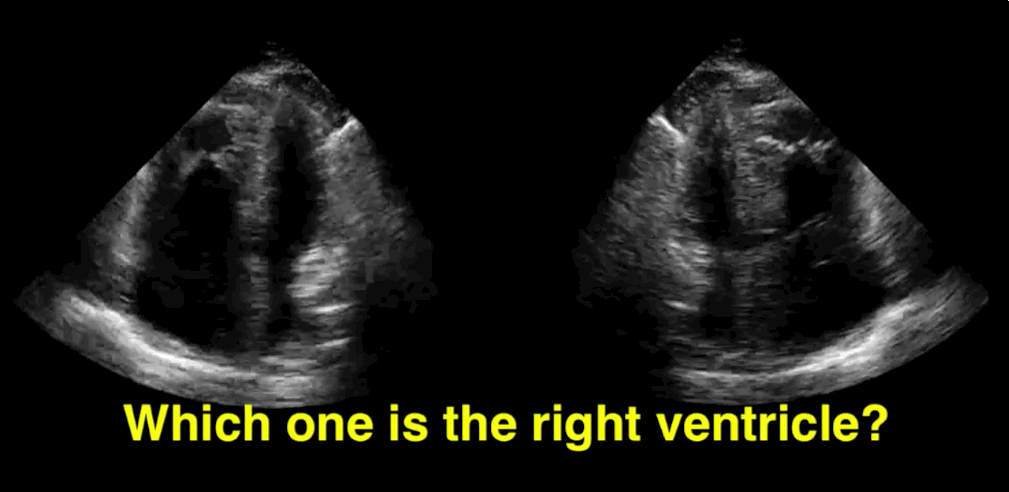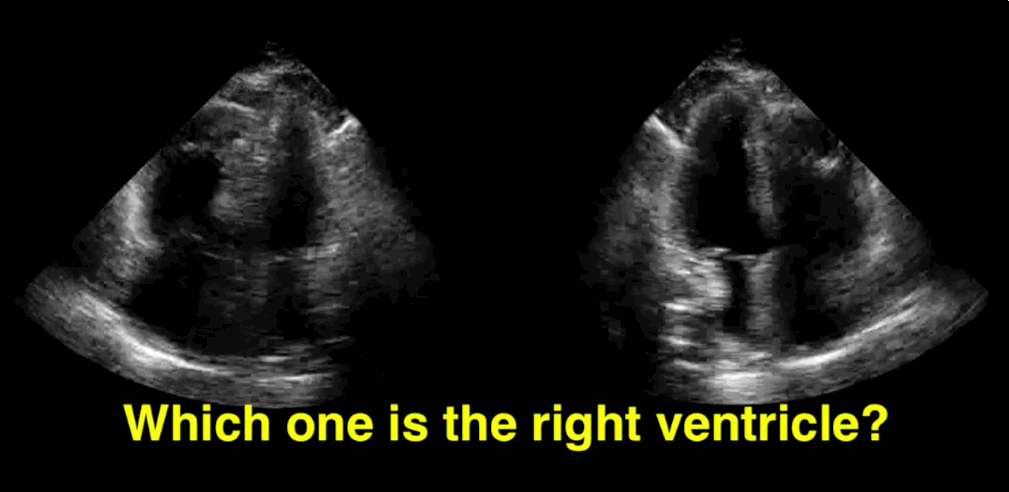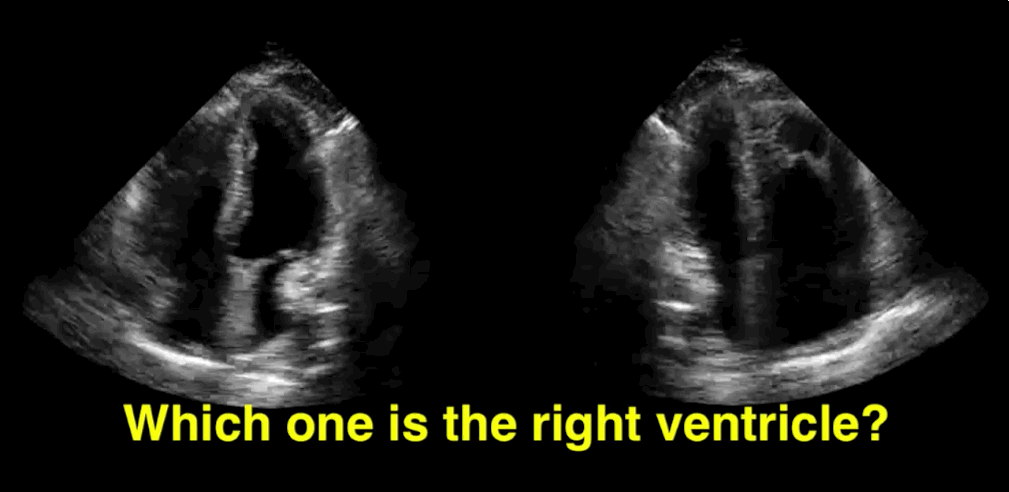
Tricks to distinguish the RV from LV in apical four-chamber view
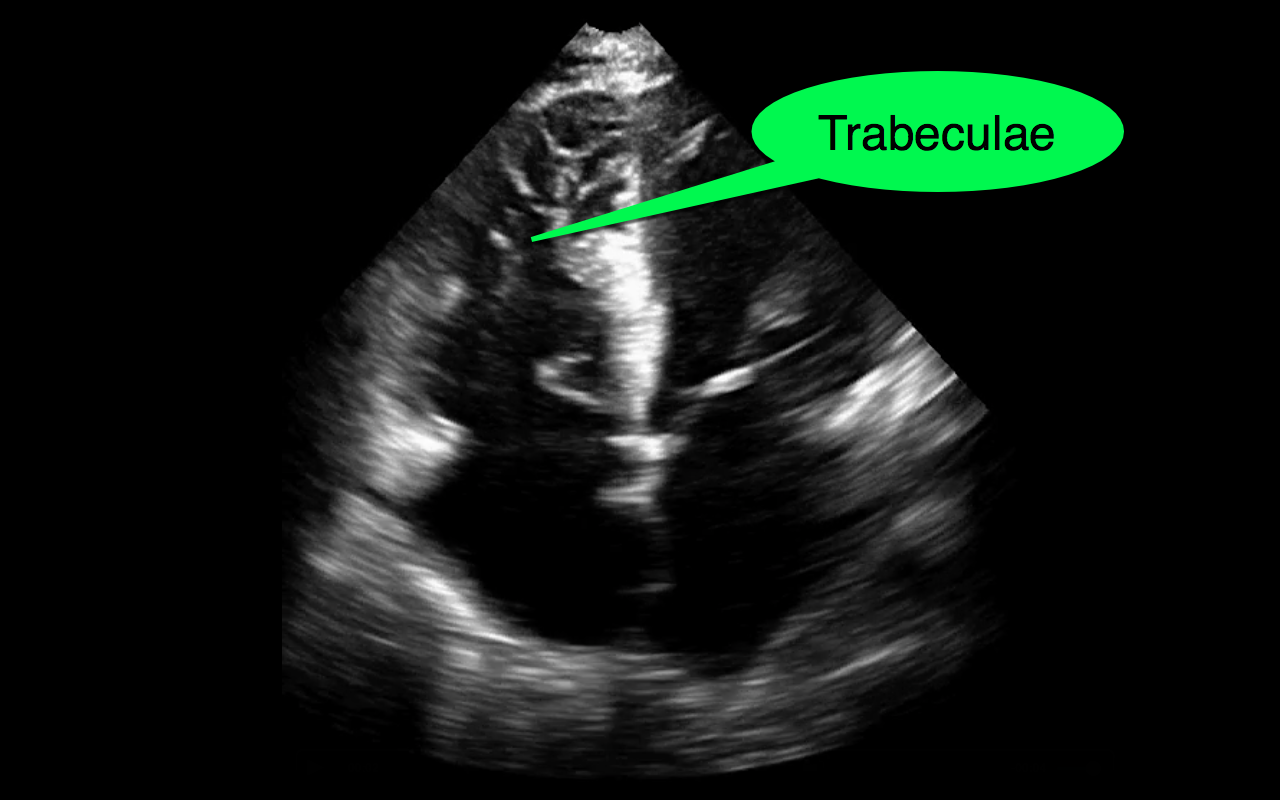
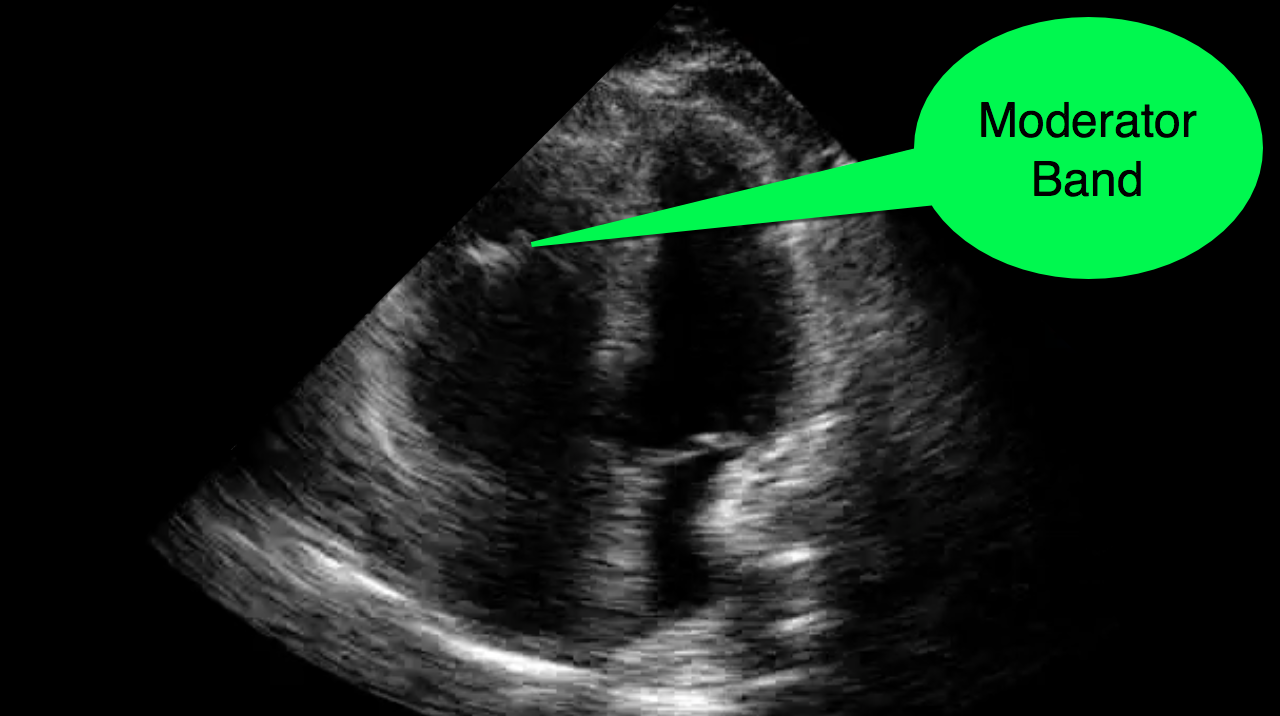
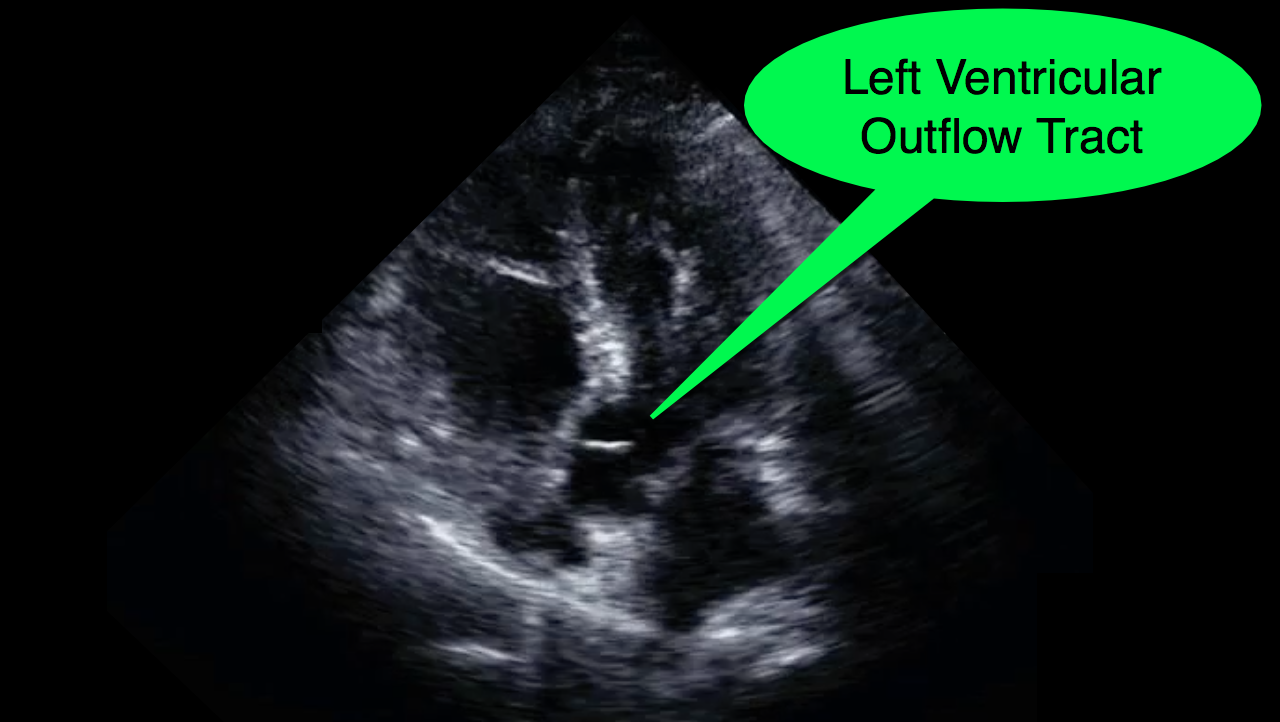
Follow me on Twitter (@criticalcarenow) or Google+ (+criticalcarenow)
Category: Critical Care
Keywords: NIPPV (PubMed Search)
Posted: 4/7/2015 by Feras Khan, MD
Click here to contact Feras Khan, MD
Cabrini L et al. Noninvasive ventilation and survival in acute care settings: a comprehensive systematic review and metaanalysis of randomized controlled trials. Crit Care Med 2015; 43:880-888.
Category: Critical Care
Posted: 3/31/2015 by Mike Winters, MBA, MD
Click here to contact Mike Winters, MBA, MD
Mechanical Ventilation in the ED
Category: Critical Care
Keywords: mechanical ventilation, ARDS, PEEP (PubMed Search)
Posted: 3/24/2015 by John Greenwood, MD
(Updated: 5/13/2026)
Click here to contact John Greenwood, MD
Stop looking for the “Best PEEP”, aim for a “Better PEEP”
Mechanical ventilation settings in the patient with acute respiratory distress syndrome (ARDS) need to provide adequate gas exchange and prevent ventilator induced lung injury (VILI). Positive end-expiratory pressure (PEEP) is often prescribed with consideration of the patient’s FiO2 requirement, estimated chest wall compliance, and hemodynamic tolerance.
So what is the best strategy for PEEP prescription?
In a recent review, Gattinoni & colleagues analyzed a number of the recent studies examining PEEP optimization. In this paper, the authors conclude that there is no “Best PEEP,” and regardless of the level chosen there will be some degree of intratidal recruitment-derecruitment and VILI. They go on to recommend a PEEP prescription strategy that reflects the severity of ARDS using the patient’s PaO2/FiO2 or P/F ratio.
Bottom line: There is no “Best PEEP” however, a “Better PEEP” is one that is primarily tailored to the severity of the patient’s ARDS, but also compensates for chest wall resistance and minimizes hemodynamic compromise.
References
Follow me on Twitter @JohnGreenwoodMD
Category: Critical Care
Posted: 3/17/2015 by Haney Mallemat, MD
(Updated: 3/18/2015)
Click here to contact Haney Mallemat, MD
The results of a multi-center trial from the UK, the ProMISe trial, were just released and it confirms what two prior studies (i.e., ProCESS and ARISE) have already shown; there does not appear to be any difference in mortality when septic patients are treated with a strategy of early-goal directed therapy as compared to usual care.
Patients were included in the ProMISe trial if they were in septic shock and were then randomized to either the EGDT group (630 patients) or the usual care group (630 patients); a total of 1,260.
The primary end-point was all cause mortality at 90 days and there was no difference shown in the primary outcome. There were no differences found in the measured secondary outcomes (e.g., serious adverse events)
This trial adds to the evidence that septic patients may not benefit from protocolized (i.e., EGDT) care versus usual care. One explaination why, is that our "usual care" in 2015 has significantly changed since the introduction of EGDT in 2001.
Follow me on Twitter (@criticalcarenow) or Google+ (+criticalcarenow)
http://www.nejm.org/doi/pdf/10.1056/NEJMoa1500896
Category: Critical Care
Keywords: massive transfusion, trauma, bleeding, critical care, severe trauma, PROPPR (PubMed Search)
Posted: 3/10/2015 by Feras Khan, MD
Click here to contact Feras Khan, MD
Transfusion in Major Trauma: The PROPPR Trial
What should we be transfusing in major trauma?
The Trial
Results
Conclusions
How does this affect my practice?
A 1:1:1 transfusion practice is safe and can decrease mortality from hemorrhage in major trauma
Other points: control bleeding, permissive hypotension, avoid crystalloids, use TEG to guide therapy (TXA etc)
Category: Critical Care
Posted: 3/3/2015 by Mike Winters, MBA, MD
Click here to contact Mike Winters, MBA, MD
High-Flow Nasal Cannula for Apneic Oxygenation
Miguel-Montanes R, et al. Use of high-flow nasal cannula oxygen therapy to prevent desaturation during tracheal intubation of intensive care patients with mild-to-moderate hypoxemia. Crit Care Med 2015;43:574-83.
Category: Critical Care
Keywords: CVP (PubMed Search)
Posted: 2/24/2015 by John Greenwood, MD
Click here to contact John Greenwood, MD
The Role of the CVP in a Post- “7 Mares” Era
The role for using central venous pressure (CVP) as a measure of volume responsiveness has largely fallen out of favor over the years.1 There are certainly better indices for fluid responsiveness, but don’t be fooled – the CVP isn’t a one trick pony. In fact, a high or rapidly rising CVP should raise a significant concern for impending cardiovascular collapse.
Consider the following differential diagnosis in the patient with an abnormally high or rising CVP ( >10 cm H2O).
Bottom Line: In a time where the utility of the CVP has been largely dismissed, remember that an abnormal CVP offers great deal of information beyond a simple measure of volume status.
References
Follow me on Twitter: @JohnGreenwoodMD
Category: Critical Care
Posted: 2/17/2015 by Haney Mallemat, MD
Click here to contact Haney Mallemat, MD
As the cold and snow rips through the United States, hypothermia is a major concern because each year approximately 1,300 Americans die of hypothermia.
Classification of hypothermia:
The risk of cardiac arrest increases when the core temperature is less than 32 Celsius and significantly rises when the temperature is less than 28 Celsius. Rapid rewarming is required as part of resuscitation should cardiac arrest occur.
A rescue therapy to consider (when available) is extra corporeal membrane oxygenation (ECMO). ECMO not only provides circulatory support for patients in cardiac arrest, but allows re-warming of patients by 8-12 Celsius per hour.
Some studies quote survival rates of 50% with hypothermic cardiac arrest patients receiving ECMO versus 10% in similar patients who do not receive ECMO.
As winter lingers in the United States, consider speaking to your cardiac surgeons now to plan an Emergency Department protocol for hypothermic patients that may require ECMO.
Follow me on Twitter (@criticalcarenow) or Google+ (+criticalcarenow)
Category: Critical Care
Keywords: VAP, chlorhexidine baths, subglottic suctioning (PubMed Search)
Posted: 2/10/2015 by Feras Khan, MD
Click here to contact Feras Khan, MD
Updates in preventative strategies in the ICU
Preventing Ventilator Associated Pneumonia (VAP)
The trial
Bottom Line
Daily bathing with chlorhexidine does not reduce health care associated infections
The trial
Bottom Line
Category: Critical Care
Posted: 2/3/2015 by Mike Winters, MBA, MD
Click here to contact Mike Winters, MBA, MD
Hypertensive Emergency Pearls
Monnet X, Marik PE. What's new with hypertensive crisis? Intensive Care Med 2015; 41:127-130.
Category: Critical Care
Keywords: Methanol, toxicology, methanol toxicity, critical care (PubMed Search)
Posted: 1/20/2015 by John Greenwood, MD
(Updated: 1/30/2015)
Click here to contact John Greenwood, MD
Extracorporeal Treatment Strategies for Acute Methanol Poisoning (When to Dialyze)
Methanol toxicity is classically included in the differential for the intoxicated patient presenting to the ED. Add a negative EtOH level, anion/osmolar gap, blindness and you have yourself a slam dunk diagnosis. The goal is to stop the liver from metabolizing methanol to formic acid. Outside of fomepizole (or old school ethanol therapy), dialysis is often discussed, but when should you actually get the nephrologist on the phone?
This month the Extracorporeal Treatments in Poisoning Workgroup released a systematic review and consensus statement to help clinicians decide when to pull the HD trigger. Their suggestions are below.
When to start HD:
Which Modality: Intermittent HD (IHD) should be used over continuous renal replacement therapies (CRRT), as you can clear the toxin faster with higher HD flows.
When to stop HD: Extracorporeal treatment can be terminated when the methanol concentration is less than 200 mg/L or 6.2 mmol/L and a clinical improvement is observed.
Bottom Line: Consider early hemodialysis in most patients presenting with methanol toxicity. Clinical exam and routine lab testing will likely provide enough information to determine the need for IHD, but specific methanol levels can be helpful to guide adjunctive treatment options.
Reference
Roberts DM, Yates C, Megarbane B, et al. Recommendations for the Role of Extracorporeal Treatments in the Management of Acute Methanol Poisoning: A Systematic Review and Consensus Statement. Crit Care Med. 2015;43(2):461-472.
Follow me on Twitter @JohnGreenwoodMD
Category: Critical Care
Posted: 1/20/2015 by Haney Mallemat, MD
Click here to contact Haney Mallemat, MD
Follow me on Twitter (@criticalcarenow) or Google+ (+criticalcarenow)
Category: Critical Care
Keywords: diaphragm weakness, respiratory failure (PubMed Search)
Posted: 1/13/2015 by Feras Khan, MD
Click here to contact Feras Khan, MD
Diaphragm weakness and its significance
There are several ways to monitor diaphragm strength and function
Clinical Relevance
Curr Opin Crit Care. 2015 Feb;21(1):34-41. doi: 10.1097/MCC.0000000000000168.
Monitoring and preventing diaphragm injury.
Category: Critical Care
Posted: 1/6/2015 by Mike Winters, MBA, MD
Click here to contact Mike Winters, MBA, MD
"PQRST" - Capnography in Cardiac Arrest
Heradstveit BE, Heltne JK. PQRST - A unique aide-memoire for capnography interpretation during cardiac arrest. Resuscitation 2014; 85:1619-20.
Category: Critical Care
Posted: 12/30/2014 by John Greenwood, MD
Click here to contact John Greenwood, MD
Cartoons Kill: A new high-risk patient for critical illness & death
This past month, the BMJ published an impressive retrospective review that analyzed nearly 80 years of data to find that animated characters in children’s films are in fact at a very high-risk for death when compared to characters in adult dramas.
Films ranged from 1937 (Snow White) to 2013 (Frozen) and were compared against the two highest gossing dramatic films in that same year. The authors found that nearly two thirds of the children’s animated films contained an on-screen death of an important character compared to only half in adult dramas.
Fatalities were most commonly the result of:
Other high-risk animated characters include the parents of the protagonist (17.8% mortality) and nemeses (28.9% mortality). Median survival time was approximately 90 minutes (much less than the usual ED LOS!)
Notable early on-screen deaths included Nemo’s mother being eaten by a barracuda 4 minutes into Finding Nemo, Tarzan’s parents being killed by a leopard 4 minutes into Tarzan, and Cecil Gaines’ father being shot in front of him 6 minutes into The Butler.
The author’s intention was to point out the psychological impact of death on young children, but I think the authors also highlight an important, high-risk patient population that could present to your ED.
Bottom Line: Animated characters should be aggressively resuscitated and strongly considered for admission to a higher level of care should they present to your ED, as they appear to be at high-risk for death and rapid decompensation.
May all of you have a happy and safe 2015!
Reference
1. Colman I, Kingsbury M, Weeks M, et al. CARTOONS KILL: casualties in animated recreational theater in an objective observational new study of kids' introduction to loss of life. BMJ. 2014;349:g7184.
Follow me on Twitter: @JohnGreenwoodMD
Category: Critical Care
Posted: 12/23/2014 by Haney Mallemat, MD
Click here to contact Haney Mallemat, MD
Treating ischemic strokes with interventional therapies (e.g., clot retrievers, stents, intra-arterial tPA, etc.) is nothing new, but there has never been a randomized control trial demonstrating benefit until recently.
The prospective MR CLEAN trial evaluated whether interventional therapies (i.e., either mechanical intervention or intra-arterial tPA) would confer benefit; patients were included if there was an acute occlusion within the proximal intracranial portion of the anterior cerebral circulation.
90% of patients received alteplase prior to randomization; there were 233 patients in the intervention group (alteplase + intraarterial intervention) and 267 patients in the usual care care arm (alteplase only); all patients were treated within 6 hours of symptoms onset
The primary outcome was functional independence at 90 days; an absolute difference of 13.5 percentage points favoring the intervention group was found. There were no significant differences in mortality or symptomatic intracerebral hemorrhage.
Despite these exciting results, we must pause and ask why this was this the first randomized trial demonstrating benefit when previous trials could not? Here are three blogs posts that deep dive this question and raise even more questions:
Follow me on Twitter (@criticalcarenow) or Google+ (+criticalcarenow)
